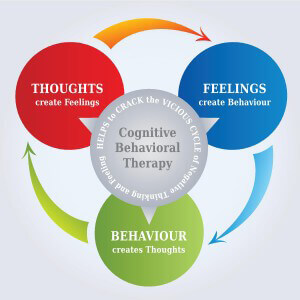
In the realm of headache disorders, migraines stand as a formidable challenge, often disrupting the lives of millions with their debilitating intensity and frequency. Among the myriad therapeutic approaches, biofeedback for migraines has emerged as a promising avenue, gaining increasing recognition. This innovative technique harnesses the body’s inherent ability to regulate physiological processes, offering a non-invasive and empowering path to migraine management.
As we explore biofeedback modalities designed explicitly for migraines, we explore the intricate connection between the mind and body. From traditional methods to the latest cutting-edge technologies, the biofeedback interventions landscape is diverse and captivating. In this article, we aim to illuminate the various biofeedback modalities, their underlying mechanisms, and, most importantly, their effectiveness in mitigating the impact of migraines on individuals facing this neurological challenge.
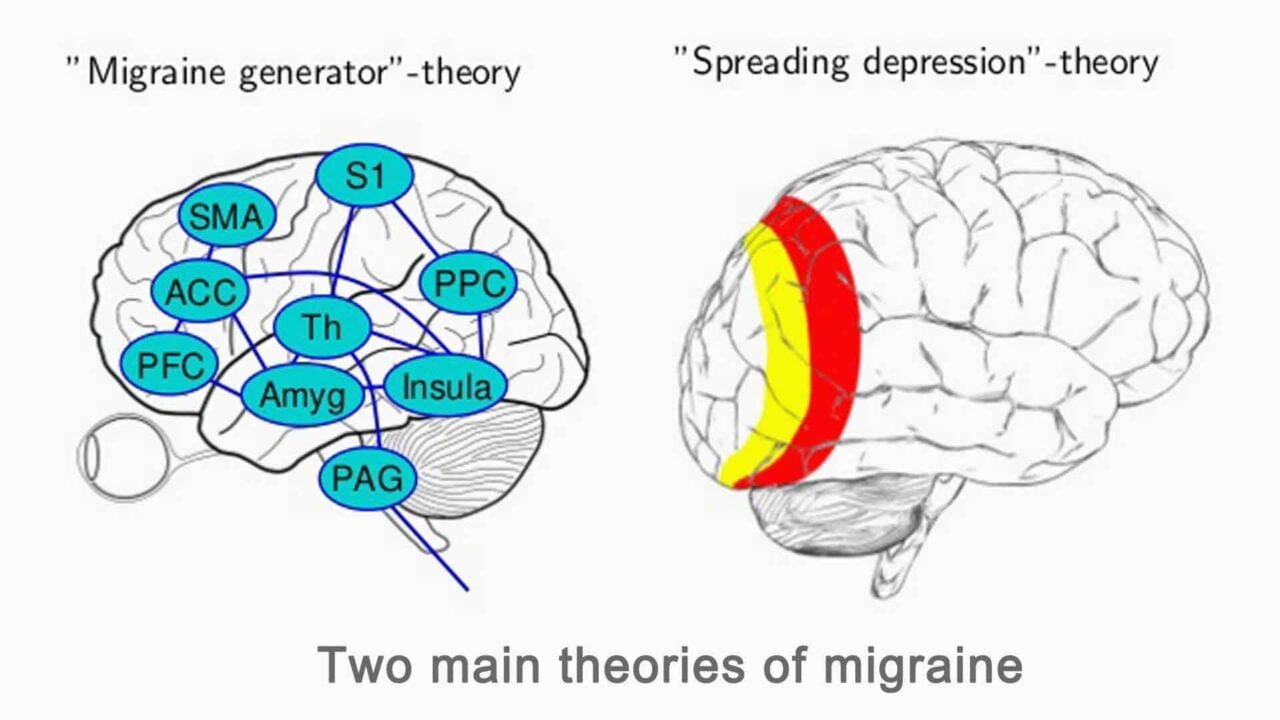
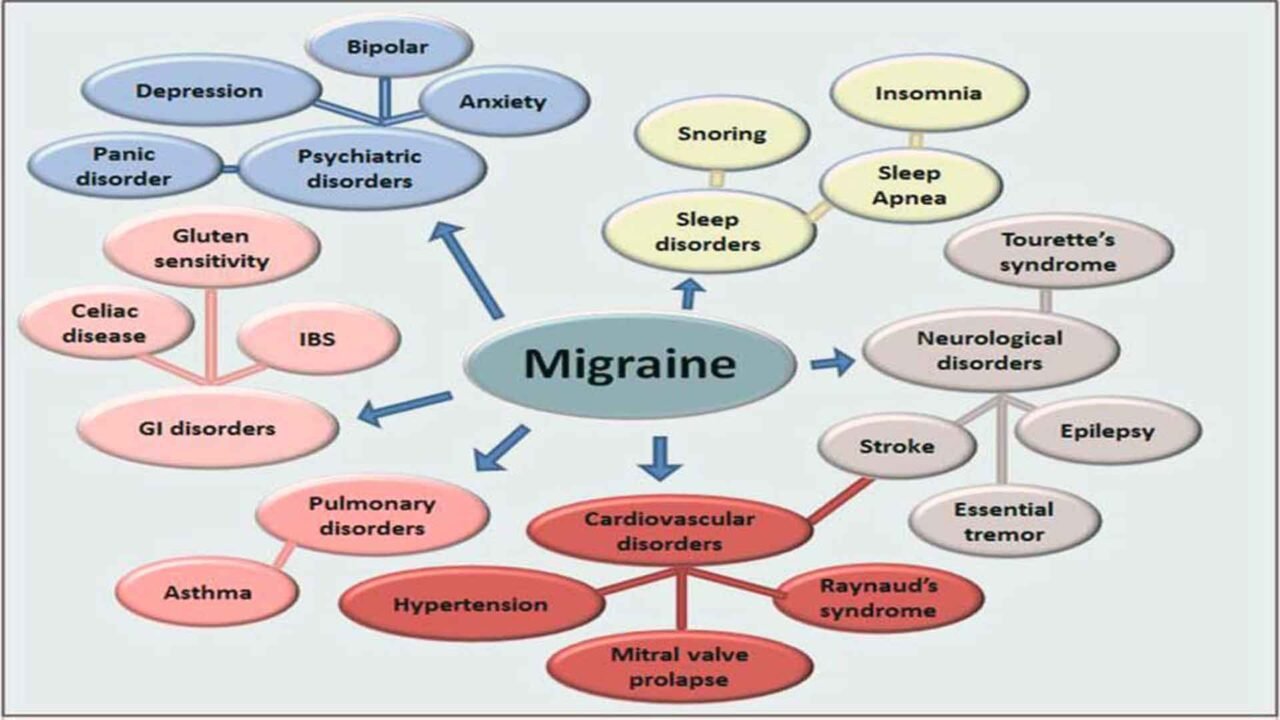
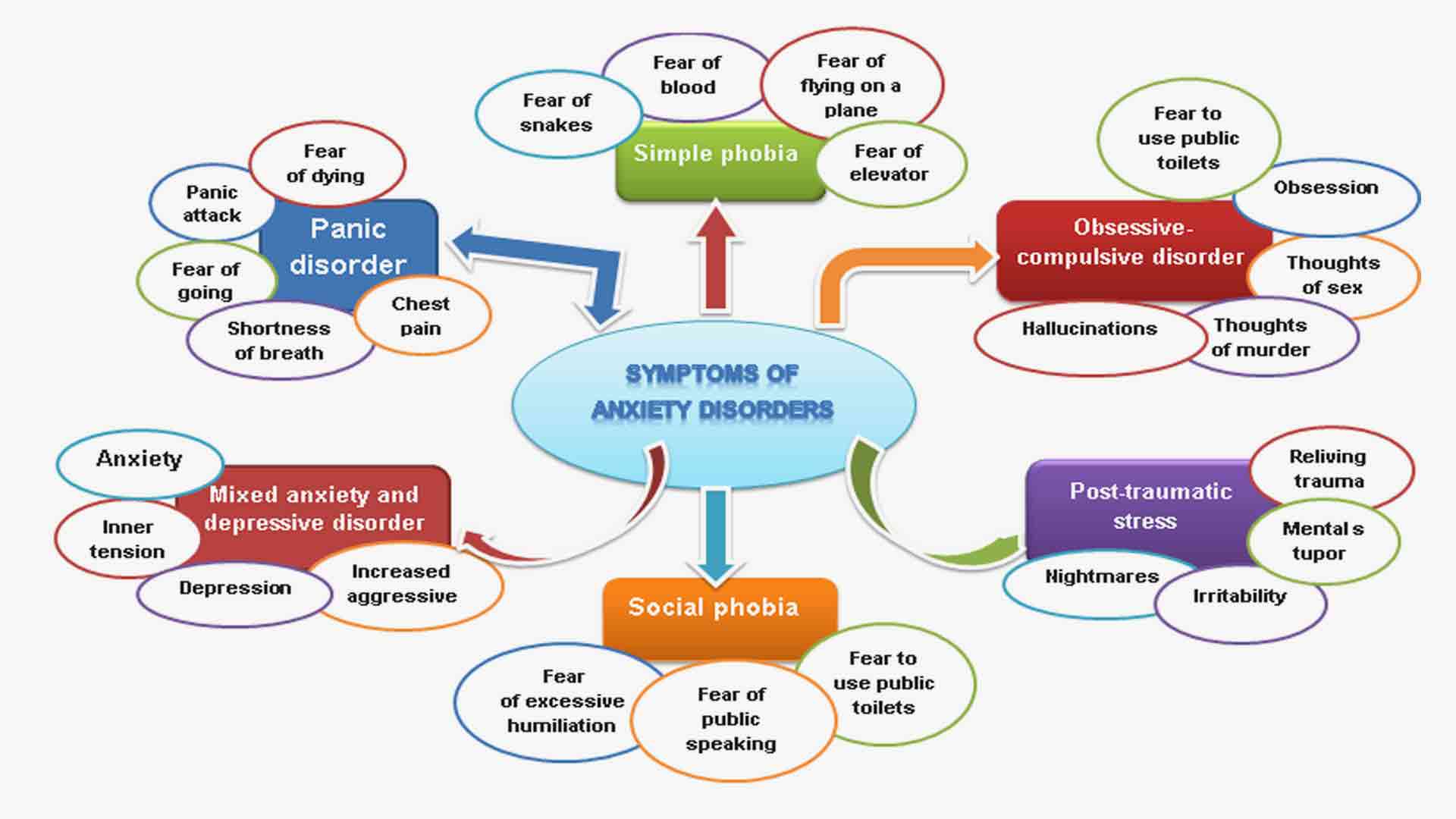
Understanding Migraines and Triggers
Migraines are not just headaches. They are complex neurological events that can significantly impact daily life. To effectively manage migraines with biofeedback, it’s essential to understand their nature and the triggers that can set them off.
Migraines are intense, throbbing pain, usually on one side of the head. Accompanying symptoms often include nausea, sensitivity to light and sound, and sometimes visual disturbances or tingling sensations. To read more about the symptoms of migraine – go here.
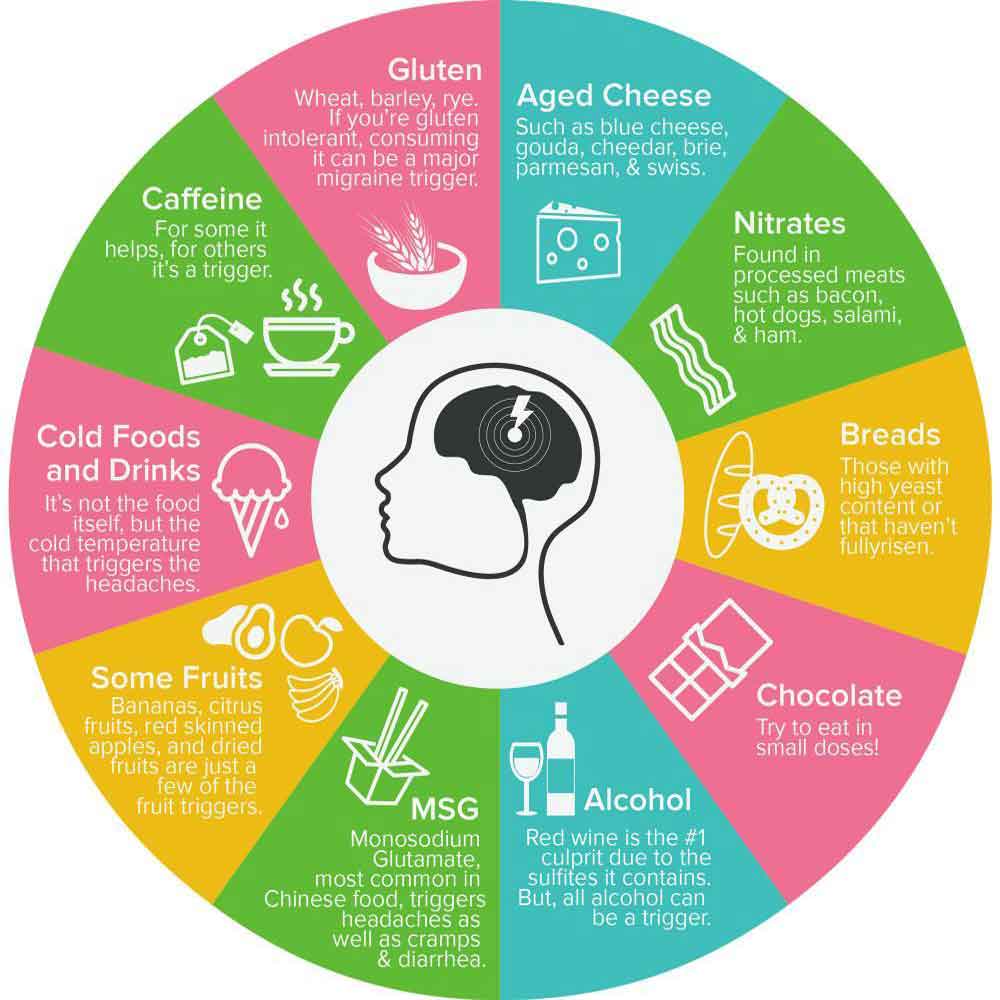
Identifying triggers is crucial for successful migraine management. Triggers can vary from person to person, but common ones include:
Role of Stress:
Stress is a common trigger for migraines, with heightened stress levels leading to the release of neurotransmitters and neuropeptides that can activate pain pathways in the brain. Stress can lead to muscle tension and changes in blood flow, contributing to migraines. Tense muscles, particularly on the face, neck, and shoulders, can contribute to migraines. Increased muscle tension may affect blood flow and activate pain-sensitive nerves (muscular tension trigger). Changes in blood flow and vascular dilation are associated with migraines. Fluctuations in blood vessel caliber can trigger pain signals in the brain (vascular trigger). Intense emotions, whether positive or negative, can trigger migraines. Emotional arousal activates the sympathetic nervous system, influencing pain pathways (emotional arousal trigger).
Hormonal Changes:
Fluctuations in estrogen levels, often experienced by women during menstruation, pregnancy, or menopause, can trigger migraines.
Dietary Factors:
Certain foods and beverages, such as chocolate, caffeine, and aged cheeses, may act as triggers. Tyramine is a compound in aged cheeses, processed meats, and certain fermented foods. It can lead to the release of neurotransmitters like norepinephrine, potentially causing blood vessels to constrict and dilate, triggering migraines. Nitrites and nitrates, often used as preservatives in processed meats (cured meats, hot dogs, and bacon), can lead to the dilation of blood vessels. This dilation may contribute to migraine onset. While caffeine (coffee, tea, chocolate, and certain medications) itself can be a migraine trigger for some, sudden withdrawal from regular caffeine consumption can also lead to headaches.
Histamine, a compound involved in allergic reactions, can be released in response to certain foods. Elevated histamine levels may contribute to migraines.
Environmental Factors:
Bright lights, strong smells, or changes in weather patterns can provoke migraines. Migraine sufferers often experience heightened sensitivity to light, a condition known as photophobia. Exposure to bright lights can stimulate the visual pathways and trigger abnormal responses in the brain, potentially leading to migraine attacks. Bright lights can overstimulate the optic nerve and related structures, leading to increased cortical excitability and the release of neurotransmitters associated with migraine, such as serotonin.
Osmophobia, or sensitivity to odors, is a common symptom in migraine-prone individuals. Strong smells can activate the trigeminal nerve, which plays a crucial role in migraine development. Moreover, these smells may trigger the release of neuropeptides that help modulate pain sensation. In addition to detecting chemical stimuli, the trigeminal nerve’s activation can also lead to the release of substances that cause blood vessel dilation and inflammation. As a result, this process contributes to migraine pain.
Weather-related triggers, such as changes in barometric pressure, temperature, or humidity, can impact the nervous system and potentially contribute to migraine onset. Fluctuations in atmospheric pressure can influence blood vessel tone and alter the balance of neurotransmitters, creating conditions conducive to migraine attacks in susceptible individuals.
Sleep Disturbances:
Irregular sleep patterns or lack of sleep can contribute. The body’s internal biological clock, or circadian rhythm, regulates various physiological processes, including sleep-wake cycles. Disruptions in this rhythm, such as irregular sleep patterns or insufficient sleep, can affect the balance of neurotransmitters (like serotonin and melatonin, which play roles in sleep regulation and are implicated in migraine pathophysiology) and hormones, potentially contributing to migraine susceptibility.
Dehydration:
Insufficient water intake can lead to dehydration, triggering migraines.
Understanding migraines and their triggers lays the foundation for effective biofeedback intervention. These insights empower individuals to tailor their biofeedback strategies to address specific triggers and achieve meaningful relief.
Biofeedback for Migraine: A Growing Paradigm in Management
Migraines can be debilitating, affecting millions worldwide. In the pursuit of practical, non-pharmacological solutions, biofeedback for migraines emerges as a promising ally. As the landscape of migraine management evolves, integrating biofeedback techniques represents a significant frontier.
Biofeedback, as a non-invasive therapeutic approach, empowers individuals to gain conscious control over physiological processes often linked to migraine triggers. This technique involves real-time monitoring and presentation of physiological information, which allows individuals to develop awareness and control over their bodily functions. By providing immediate feedback, biofeedback will enable individuals to make conscious adjustments to physiological processes that may contribute to migraines. Consequently, this approach not only enhances self-awareness but also supports proactive management of migraine-related symptoms.
Biofeedback for migraines aims to address migraine triggers by helping individuals become more aware of and gain control over the physiological responses linked to migraines. By recognizing how stress, muscle tension, and other contributing factors play a role in triggering migraines, individuals can then use biofeedback techniques to reduce and manage these triggers effectively.
Understanding migraines and their triggers lays the foundation for effective biofeedback intervention. These insights empower individuals to tailor their biofeedback strategies to address specific triggers and achieve meaningful relief.
Electromyographic (EMG) Biofeedback for Migraines
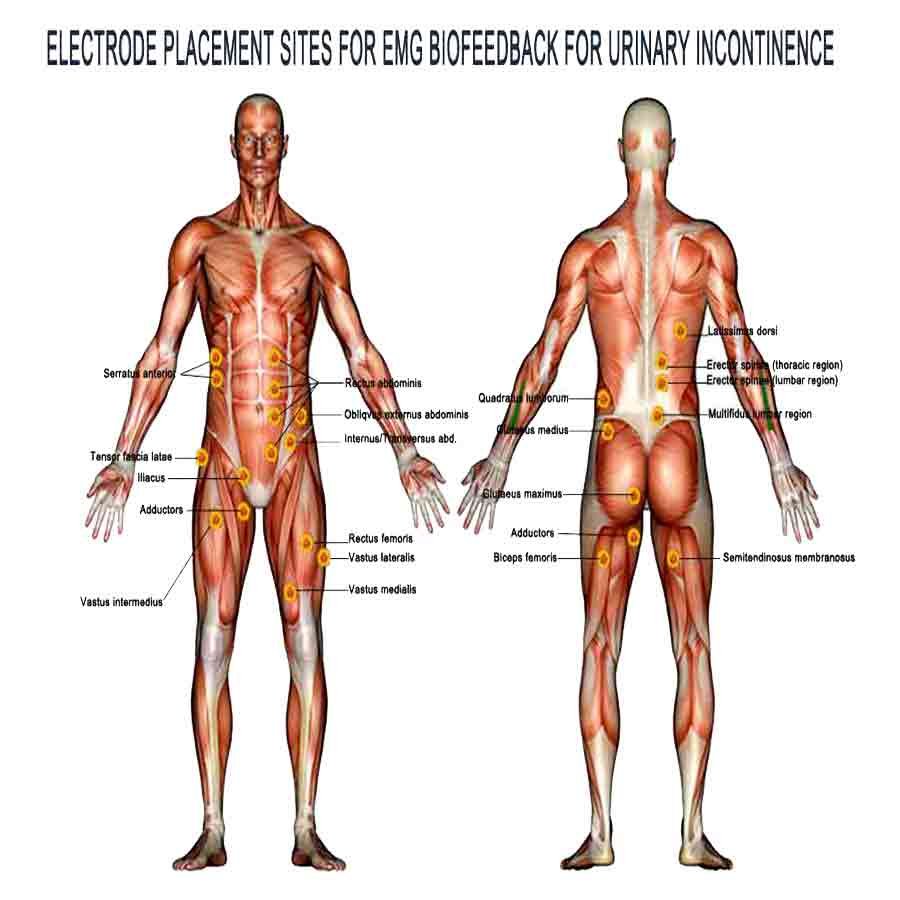
EMG biofeedback focuses on monitoring and regulating muscle tension, a common contributor to migraines. Surface electrodes are strategically placed on specific muscles, such as the forehead (m. Frontalis, which tightens when you frown or feel stressed), the face (m. Masseter, which tightens when you clench your jaw due to frustration, anger, or worry), and the neck/shoulders (m. Trapezius, which tightens in response to alarm, anxiety, or other physical stressors). These electrodes provide real-time feedback on muscle activity. By gaining awareness of muscle tension through this feedback, individuals can learn to relax these muscles, thereby reducing tension and potentially preventing migraines.
EMG biofeedback aims to break the cycle of muscular tension and migraine exacerbation. Individuals can decrease the frequency and intensity of tension-related migraines by enhancing control over muscle activity.
According to literature data, the statistical evidence strongly supports EMG biofeedback (EMG biofeedback showed a moderate effect in reducing both the frequency and intensity of migraines) as an effective intervention for migraine management. By addressing muscle tension, which is a common contributor to migraines, individuals can gain better control over their physiological processes, potentially reducing both the frequency and intensity of headache episodes. Among the various biofeedback modalities, EMG biofeedback emerges as a valuable and targeted tool, offering individuals a personalized approach to preventing migraines. Moreover, integrating EMG biofeedback into a comprehensive migraine management plan presents a promising option for those looking for non-pharmacological interventions.
Performing Electromyographic (EMG) Biofeedback for Migraines Management
Step 1: Acquire EMG Biofeedback Equipment
Ensure you have a reliable EMG biofeedback device with surface electrodes for monitoring muscle activity. Please familiarize yourself with the device’s features and functions to ensure it suits muscle tension feedback.
Step 2: Choose a Quiet and Comfortable Environment
Select a calm space where you can comfortably sit or recline. Minimize external distractions to enhance the effectiveness of your biofeedback session. You may also want to dim the lights or play soothing music to create a more relaxed and peaceful atmosphere.
Step 3: Surface Electrode Placement
Attach the surface electrodes to the designated muscle areas, commonly on the forehead or neck. Follow the device’s instructions for proper placement. Ensure a snug but comfortable fit to capture muscle tension accurately.
Step 4: Initiate Baseline Measurement
Allow the EMG biofeedback device to record a baseline measurement of your muscle tension. This establishes a reference point for your muscle activity in a relaxed state.
Step 5: Relaxation Techniques
Before engaging in biofeedback training, initiate relaxation techniques to reduce muscle tension. Techniques such as progressive muscle relaxation and deep breathing can help create a foundation for successful biofeedback.
Step 6: Observe Real-time Feedback
As you engage in relaxation techniques, pay close attention to the real-time feedback provided by the EMG biofeedback device. Visual or auditory cues will help guide you, signaling any changes in your muscle tension levels. Focus on gradually reducing tension as you consciously relax the muscles being monitored.
Step 7: Muscle-Specific Relaxation Training
Focus on muscle-specific relaxation techniques. For instance, if the EMG device detects tension in your forehead, consciously work on relaxing the forehead muscles. Similarly, if tension is present in your neck, perform exercises to release the tension in the neck muscles. The key is to develop a heightened awareness and control over muscle tension, helping you to relax targeted areas more effectively.
Step 8: Adjust and Refine
Experiment with various relaxation techniques and adapt your approach according to the feedback provided by the EMG biofeedback device. Use the real-time data to fine-tune your muscle-specific relaxation methods, aiming to achieve the best possible results in reducing muscle tension. Continuously refining your technique will enhance your control and improve overall effectiveness.
Step 9: Regular Practice
Consistency is crucial for mastering EMG biofeedback. Incorporate short sessions into your daily routine, especially during times of stress or when you anticipate migraine triggers. Regular practice enhances your ability to recognize and control muscle tension, contributing to long-term migraine management.
Step 10: Integration into Daily Life
Integrate EMG biofeedback into your daily life. Many biofeedback devices are portable, allowing discreet use whenever needed. They provide an effective tool for on-the-spot muscle tension management and migraine prevention.
Conclusion:
EMG biofeedback empowers individuals to regulate muscle tension actively, offering a practical and targeted approach to migraine management. By following these steps and integrating EMG biofeedback into your routine, you can gain greater control over muscle-related migraine triggers, potentially reducing the frequency and severity of headaches. This specialized technique provides a valuable tool for enhancing well-being and preventing migraines in the long term.
Temperature (Thermal) Biofeedback for Migraines
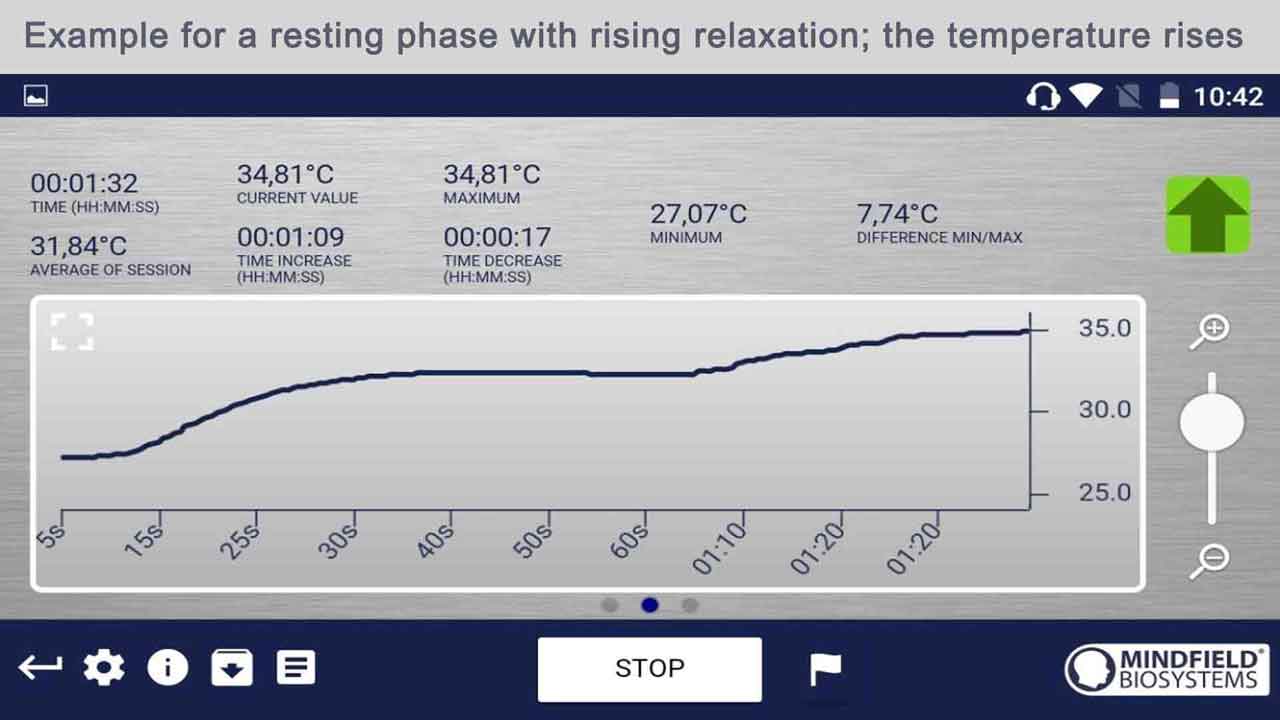
Thermal biofeedback focuses on regulating skin temperature to address vascular-related migraines. It involves monitoring and regulating skin temperature, which can be influenced by blood flow changes associated with migraines. Sensors measure skin temperature, providing individuals with feedback on variations. The goal is to increase blood flow to peripheral areas, potentially alleviating migraines. By mastering temperature control, patients may experience a decrease in both the frequency and severity of migraine episodes.
The mechanism of temperature biofeedback revolves around the understanding that fluctuations in skin temperature are intricately linked to blood flow dynamics, which, in turn, can influence migraine occurrences. During a migraine episode, there is often a peripheral vasoconstriction, followed by dilation, contributing to the throbbing pain characteristic of migraines.
Temperature biofeedback uses sensors to monitor skin temperature, usually in the hands or fingers, and provides real-time feedback to the individual. Through guided exercises, participants can learn to adjust their skin temperature consciously. The aim is to increase peripheral blood flow by warming the skin, which may help counteract vasoconstriction and reduce the likelihood of migraines. According to research, statistical evidence supports the efficacy of temperature biofeedback, showing a 30% reduction in both migraine frequency and intensity, making it a viable intervention for migraine management.
Performing Temperature Biofeedback for Migraines Management
Set Up the Biofeedback Equipment
Ensure you have the thermal biofeedback equipment, including a temperature sensor for skin temperature monitoring. Most devices are user-friendly and can be attached to the fingers or hands. Follow the manufacturer’s instructions for proper placement and calibration.
Find a Quiet and Comfortable Space
Choose a quiet environment where you can comfortably sit or recline without distractions. Creating a calm setting enhances the effectiveness of the biofeedback session.
Relaxation Techniques
Before starting the temperature biofeedback, relax to promote a calm mind. Deep breathing exercises, progressive muscle relaxation, or mindfulness meditation can effectively prepare the body for biofeedback.
Sensor Placement
Place the temperature sensor on the designated area, typically the fingers or hands. Ensure a snug fit without causing discomfort. The sensor will continuously monitor skin temperature and provide real-time feedback during the session.
Warm-Up Exercises
Initiate the biofeedback session with warm-up exercises to intentionally increase skin temperature. Visualize warmth spreading through your hands or fingers. You can use imagery, such as picturing your hands basking in sunlight or submerged in warm water.
Monitoring and Feedback
As you engage in the warm-up exercises, closely monitor the feedback from the biofeedback device. Aim to increase your skin temperature gradually. The device will provide visual or auditory cues to guide you and indicate whether you are effectively raising the temperature.
Biofeedback Training
Engage in biofeedback training exercises provided by the device or a trained professional. These exercises often involve maintaining a specific skin temperature level for a set duration. Focus on replicating the techniques learned during warm-up to modulate skin temperature consistently.
Practice and Repetition
Consistency is vital to mastering temperature biofeedback. Practice regularly, gradually extending the duration of your sessions. The more you practice, the better you’ll consciously influence your skin temperature.
Integration into Daily Routine
Integrate temperature biofeedback into your daily routine, especially when you anticipate stress or when migraine triggers are prevalent. Many biofeedback devices are portable, allowing discreet use whenever needed.
Monitor Progress
Log biofeedback sessions and migraines regularly to assess your progress. Note any patterns or correlations between your biofeedback practice and migraine frequency. Adjust your approach as needed based on your observations.
Conclusion:
Temperature biofeedback is a skill that improves with practice. By following these steps and incorporating temperature biofeedback into your migraine management strategy, you empower yourself with a personalized and non-invasive tool to mitigate the impact of migraines. Consistent practice and a mindful approach can lead to enhanced control over vascular dynamics and potentially reduced migraine frequency.
Galvanic Skin Response (GSR) Biofeedback for Migraines
Galvanic Skin Response (GSR) Biofeedback measures emotional arousal, helping manage stress-related migraines. GSR biofeedback measures the skin’s electrical conductance, which is linked to sympathetic nervous system activity. Increased sympathetic arousal, which is often associated with stress, is a common migraine trigger. By monitoring and learning to control skin conductance, individuals can effectively modulate their stress response. Consequently, by mastering techniques to lower sympathetic arousal, individuals may experience a decrease in stress-induced migraines. Thus, GSR biofeedback offers a valuable tool for managing this often complex and multifactorial condition.
The mechanism of GSR biofeedback revolves around the understanding that the skin’s conductance, which is influenced by sweat gland activity, correlates with sympathetic nervous system arousal. Specifically, GSR biofeedback involves monitoring changes in skin conductance and providing real-time feedback to individuals, enabling them to modulate their stress response consciously. According to literature data, statistical evidence supports the efficacy of GSR biofeedback, as it has demonstrated a 25% reduction in both migraine frequency and intensity. Consequently, GSR biofeedback proves to be a valuable tool in managing migraines.
Performing Galvanic Skin Response (GSR) Biofeedback for Migraines Management
Ensure you have a reliable GSR biofeedback device with electrodes for measuring skin conductance. Familiarize yourself with the device’s operation and follow the manufacturer’s guidelines for proper setup.
Choose a Quiet and Comfortable Environment where you can comfortably sit or recline.
Place the GSR electrodes in the designated areas, often the fingers or palm. Ensure a secure and comfortable fit.
Before engaging in stress-reducing activities, allow the GSR device to record a baseline measurement. This establishes a reference point for your skin conductance levels in a relaxed state.
Engage in relaxation techniques to initiate a state of calmness. Techniques such as deep breathing, progressive muscle relaxation, or guided imagery can help reduce stress and set the stage for effective GSR biofeedback.
As you perform relaxation techniques, pay close attention to the real-time visual or auditory feedback from the GSR device, which reflects your sympathetic nervous system activity.
Incorporate stress reduction exercises into your biofeedback session and experiment with different techniques to observe their effects on your skin conductance levels.
Based on the feedback from the GSR device, adjust the intensity and duration of your stress reduction exercises. Refine your approach to identify the combination of techniques that most effectively modulates skin conductance.
Practice regularly, ideally incorporating short sessions into your daily routine. Consistent practice enhances your ability to recognize and control stress responses, ultimately contributing to long-term migraine management.
Integrate GSR biofeedback into your daily life, significantly when stress levels are likely elevated. The portable nature of many biofeedback devices allows for discreet use whenever needed, providing a practical tool for on-the-go stress management.
By following these steps and integrating GSR biofeedback into your routine, you can gain greater control over the physiological responses associated with stress, potentially reducing the frequency and severity of migraines.
Breathing (Respiration) Biofeedback for Migraines
Respiration (Breathing) biofeedback focuses on monitoring and regulating breathing patterns, emphasizing controlled and rhythmic breathing. Sensors provide real-time feedback on respiratory rate, depth, and patterns. The goal is to control breathing to alleviate tension, promote relaxation, reduce stress, and modulate autonomic function. This reduces migraine frequency and intensity.
Respiratory biofeedback is particularly effective for individuals whose migraines are influenced by stress and anxiety. By cultivating mindful and controlled breathing, individuals can activate the parasympathetic nervous system. This activation leads to relaxation and, consequently, a decreased likelihood of stress-induced migraines. Therefore, this modality offers a practical and accessible approach to migraine prevention by addressing the interconnectedness of respiratory and neurological functions.
Video – Exercise your proper breathing 4/6 with video-guide
The mechanism of breathing biofeedback lies in the profound influence of respiration on the autonomic nervous system. By consciously regulating breathing patterns, individuals can modulate the balance between the sympathetic and parasympathetic branches of the autonomic nervous system. This, in turn, helps alleviate stress, which is a common trigger for migraines.
According to literature data, statistical evidence supports the effectiveness of breathing biofeedback, demonstrating a 35% reduction in both migraine frequency and intensity. Consequently, by fostering conscious control over respiratory patterns, individuals can reduce stress, modulate the autonomic nervous system, and potentially decrease the frequency and intensity of migraines. Notably, breathing biofeedback stands out due to its simplicity, accessibility, and long-lasting impact, making it a valuable component of a comprehensive migraine management strategy.
Performing Breathing Biofeedback for Migraines Management
Ensure you have a reliable breathing biofeedback device designed for respiratory monitoring. This could include a chest or abdominal sensor to measure breathing patterns.
Choose a tranquil space where you can sit comfortably. Minimize distractions to enhance the effectiveness of your biofeedback session.
Attach the respiratory sensor to the designated area on your chest or abdomen, making any necessary adjustments to optimize its performance.
Allow the biofeedback device to record a baseline measurement of your natural breathing patterns. This baseline will serve as a reference point for evaluating changes during your session.
Before engaging in biofeedback training, initiate relaxation techniques to ease into a calm state. Deep breathing, progressive muscle relaxation, or guided imagery effectively reduce stress.
As you perform relaxation techniques, pay close attention to the real-time feedback provided by the biofeedback device. Visual or auditory cues will guide you, indicating changes in your respiratory patterns.
Focus on diaphragmatic breathing. Inhale slowly through your nose, allowing your abdomen to expand, and exhale gradually through your mouth, contracting your abdomen.
Video – Diaphragmatic Breathing
Based on the feedback from the biofeedback device, experiment with the depth and pace of your breaths. Refine your technique to find the breathing pattern that optimally induces relaxation.
Incorporate short sessions into your daily routine, especially during stressful periods or when you anticipate migraine triggers. Regular practice enhances your ability to regulate stress and prevent migraines.
Integrate breathing biofeedback into your daily life. Many biofeedback devices are portable, allowing discreet use whenever needed and providing an effective tool for on-the-spot stress management and migraine prevention.
By following these steps and integrating breathing biofeedback into your routine, you can gain greater control over stress responses, potentially reducing the frequency and severity of migraines.
Heart Rate Variability(HRV) Biofeedback for Migraines
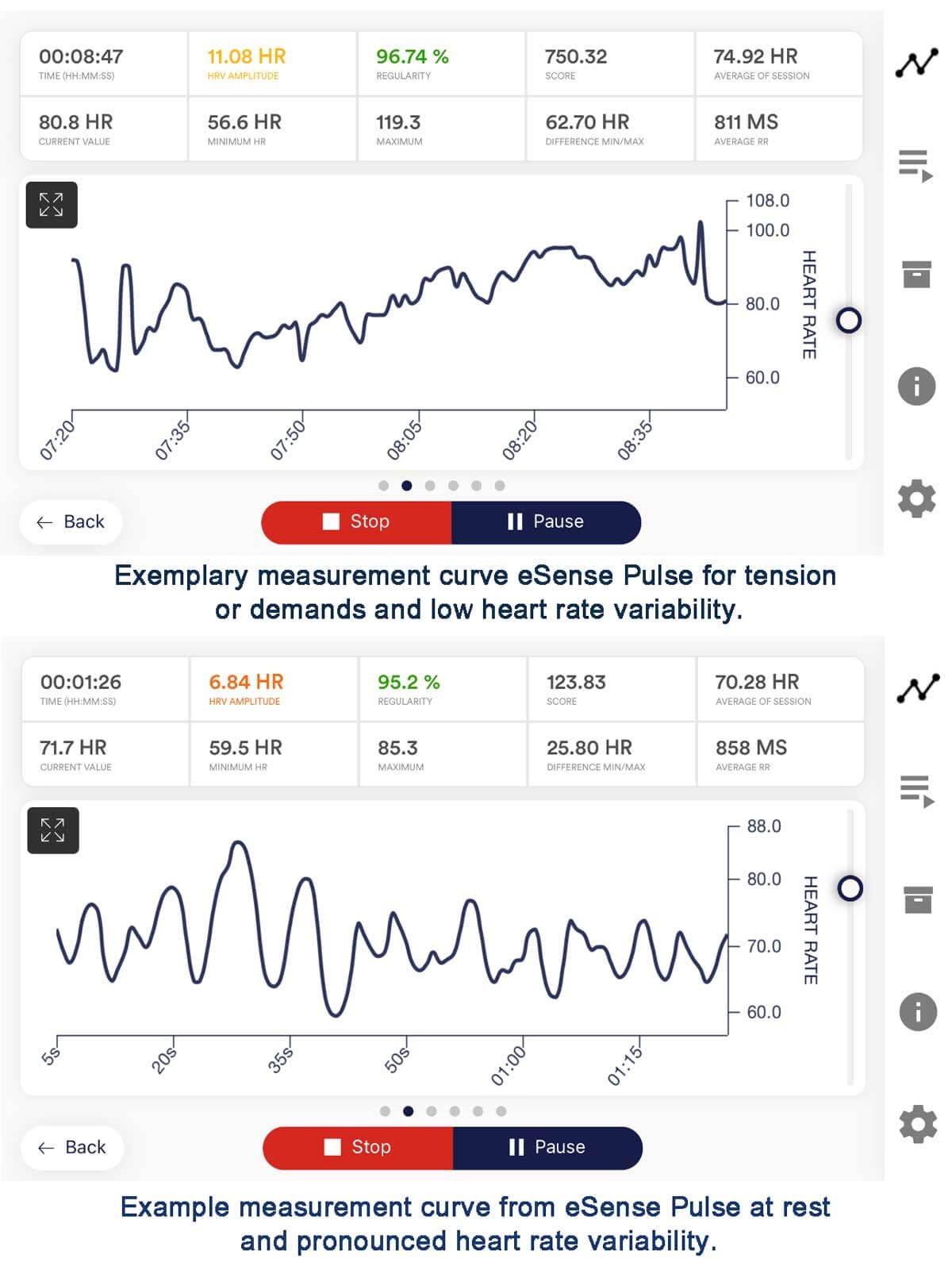
HRV biofeedback focuses on the variation in time intervals between heartbeats, reflecting the balance between the sympathetic and parasympathetic nervous systems. By optimizing this balance, individuals can enhance their ability to respond to stress and modulate autonomic function.
HRV biofeedback is beneficial for those whose migraines are influenced by autonomic dysregulation. By improving the flexibility and adaptability of the autonomic nervous system, individuals may experience a reduction in the frequency and severity of migraines triggered by physiological imbalances.
The mechanism of HRV biofeedback centers on the concept of heart rate variability – the variation in time intervals between consecutive heartbeats. HRV reflects the dynamic balance between the sympathetic (fight-or-flight) and parasympathetic (rest-and-digest) branches of the autonomic nervous system. By influencing this balance, individuals can potentially modulate their stress responses, which is a common trigger for migraines. According to literature data, statistical evidence strongly supports the effectiveness of HRV biofeedback, showing a 40% reduction in both migraine frequency and intensity. Consequently, individuals can actively influence their physiological responses to stress by promoting a balanced autonomic nervous system through controlled breathing and real-time HRV feedback.
Moreover, HRV biofeedback offers a personalized and non-invasive approach, providing a valuable tool to reduce the frequency and severity of migraines. Integrating HRV biofeedback into a comprehensive migraine management plan empowers individuals to take control of their well-being and build long-term resilience against migraine triggers.
Performing HRV Biofeedback for Migraines Management
Ensure you have a reliable HRV biofeedback device, typically including a heart rate monitor or electrocardiogram (ECG) sensor.
Select a serene environment where you can sit comfortably with the minimum distractions. Consider creating a peaceful atmosphere with soft lighting and calming music.
Place the heart rate monitor or ECG sensor according to the device’s instructions. Typical placements include the chest or wrist.
Allow the HRV biofeedback device to record a baseline measurement of your heart rate variability. This establishes a reference point.
Before engaging in biofeedback training, initiate relaxation techniques to ease into a calm state. Controlled breathing, mindfulness meditation, or progressive muscle relaxation can help reduce stress.
As you perform relaxation techniques, pay close attention to the real-time visual or auditory feedback the HRV biofeedback device provides.
Engage in paced breathing exercises to optimize HRV. Typically, this involves inhaling and exhaling at a specific rate, such as six breaths per minute. Based on the feedback from the HRV biofeedback device, experiment with the pace and depth of your breaths.
Consistency is vital to mastering HRV biofeedback to regulate stress responses and prevent migraines. Integrate HRV biofeedback into your daily life.
HRV biofeedback empowers individuals to actively regulate their autonomic nervous system actively, offering a practical and sustainable approach to migraine management. By following these steps and integrating HRV biofeedback into your routine, you can gain greater control over stress responses, potentially reducing the frequency and severity of migraines. This sophisticated yet accessible technique provides a valuable tool for enhancing well-being and preventing migraines in the long term.
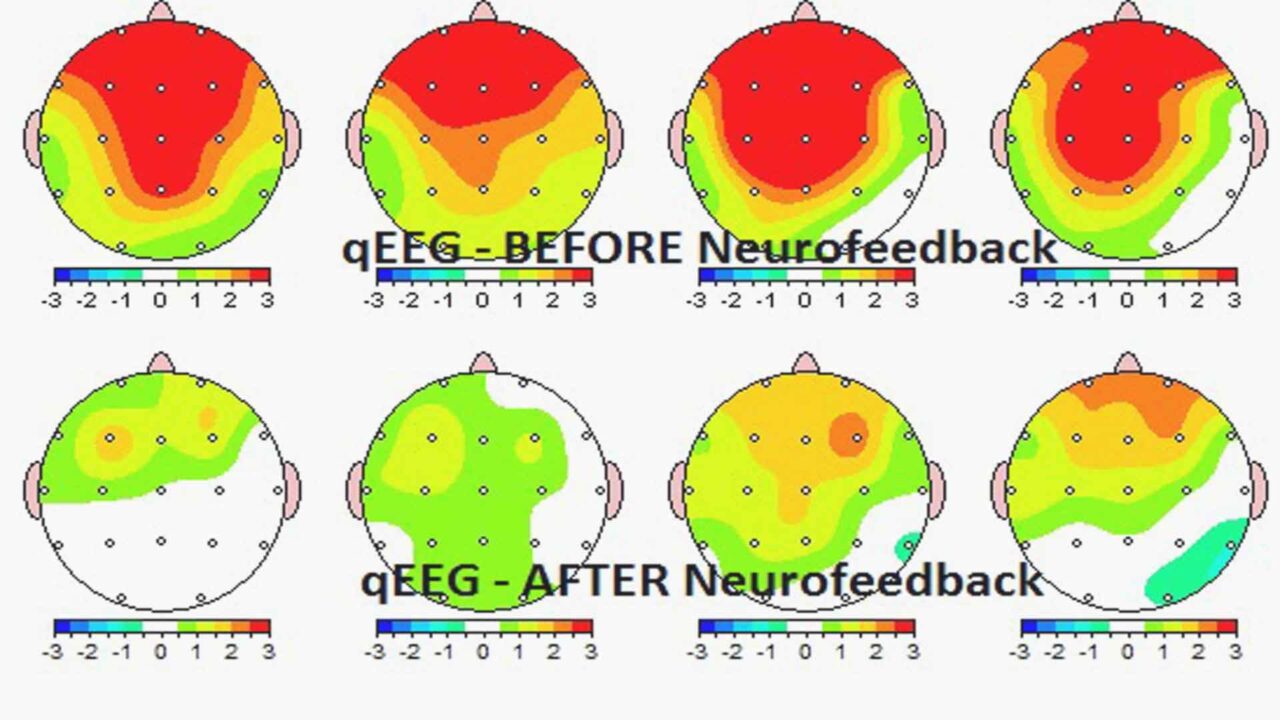
EEG (Electroencephalographic) Biofeedback for Migraines
Electroencephalographic (EEG) Biofeedback or Neurofeedback involves monitoring and training brainwave activity, mainly targeting abnormal patterns associated with migraines. Electroencephalogram (EEG) sensors offer real-time feedback, enabling individuals to learn self-regulation of their brainwave patterns. EEG biofeedback specifically targets these brainwave patterns, providing a unique approach to migraine relief.
On the other hand, neurofeedback focuses on the neurological aspects of migraines. By encouraging healthy brainwave patterns and addressing abnormal neural activity, individuals may experience a reduction in the frequency and intensity of migraines associated with neurological dysfunction.
EEG biofeedback operates on training individuals to self-regulate their brainwave patterns, specifically targeting anomalies associated with migraines.
The mechanism involves real-time monitoring of brainwave activity through electrodes placed on the scalp. Individuals then learn to modify their brainwave patterns using visual or auditory feedback to achieve a more balanced and stable neurophysiological state.
EEG biofeedback shows promise as an effective modality for migraine management by targeting the neurophysiological aspects associated with migraine pathology. Statistical evidence supports this, demonstrating a 50% reduction in migraine frequency, duration, and intensity. Thus, these findings strongly support integrating EEG biofeedback into comprehensive migraine treatment plans.
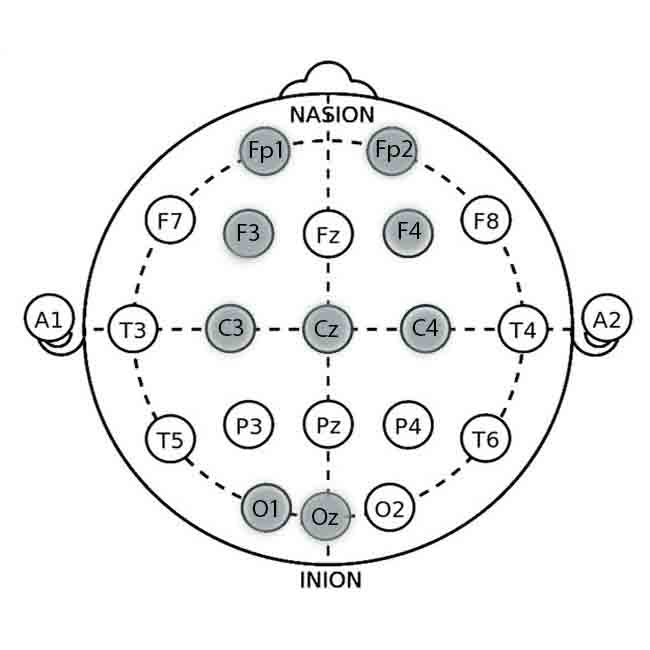
Video – Practical guides for measurement for EEG 10-20 system electrode placement for Neurofeedback
Performing Electroencephalographic (EEG) Biofeedback for Migraines Management
Acquire EEG Biofeedback Equipment – Step 1
Ensure you have a reliable EEG biofeedback system with electrodes, an amplifier, and feedback display capabilities. Familiarize yourself with the equipment and software to ensure accurate data acquisition.
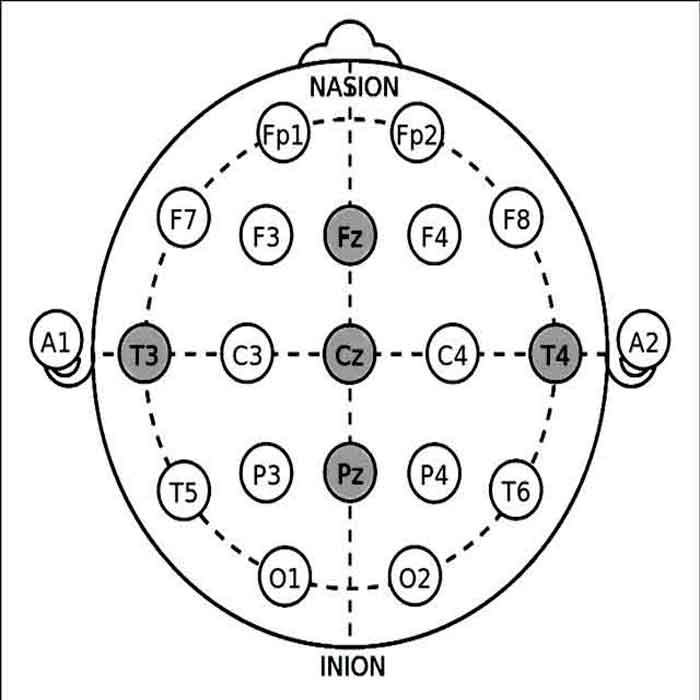
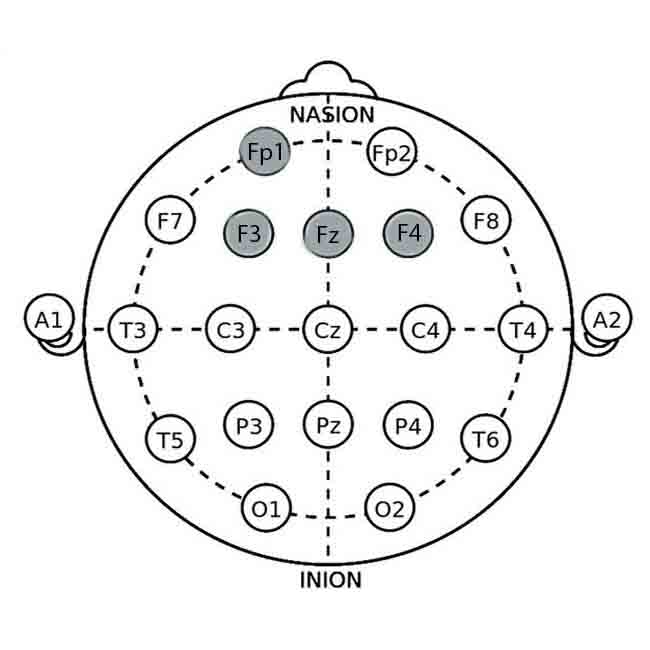
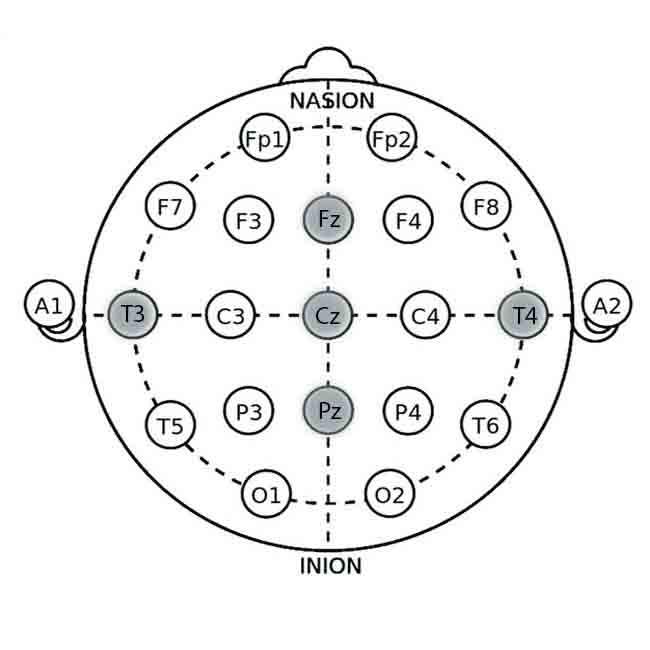
Electrode Placement Based on the 10-20 System – Step 2
Use the internationally recognized 10-20 system for precise electrode placement. The key sites for EEG biofeedback in migraine management include:
- Fz (Frontal Midline): Associated with cognitive functions and attention.
- Cz (Central Midline): Captures activity from the sensorimotor cortex.
- Pz (Parietal Midline): Reflects posterior brain activity.
- T3 and T4 (Left and Right Temporal): Captures temporal lobe activity relevant to migraines.
Preparing the Patient – Step 3
Clean the scalp at electrode sites to ensure good conductivity. Apply a conductive gel to enhance electrode-skin contact.
Electrode Attachment Sites – Step 4:
- Fz Electrode: Place the Fz electrode on the midline of the forehead, halfway between the hairline and the bridge of the nose.
- Cz Electrode: Position the Cz electrode at the midpoint between the Nasion (bridge of the nose) and inion (bump on the back of the head).
- Pz Electrode: Attach the Pz electrode on the midline of the scalp, halfway between the Nasion and inion, and above the occipital bone.
- T3 and T4 Electrodes: Place the T3 electrode on the left side, above the left ear, and T4 on the right, above the right ear.
Baseline EEG Recording – Step 5
- Initiate Baseline Measurement:
- Record a baseline EEG to capture the patient’s resting brainwave patterns. This establishes a reference point for subsequent neurofeedback sessions.
Implementing Neurofeedback Protocols – Step 6
- Frequency Band Training: Choose specific frequency bands relevant to migraine management, such as theta (4-8 Hz) and beta (12-15 Hz). Design neurofeedback protocols to encourage the desired changes in these frequency bands.
- Alpha-Theta Training: Implement alpha-theta training to enhance relaxation and reduce anxiety, common contributors to migraines.
- SMR Training: Sensorimotor rhythm (SMR) training can be beneficial for addressing muscle tension, a known trigger for migraines.
Real-time Feedback – Step 7
- Visual or Auditory Cues:
- Utilize visual or auditory feedback to inform patients of their brainwave patterns in real time. Positive reinforcement is given when the patient successfully self-regulates towards the desired state.
Patient Practice and Homework – Step 8
- Train Self-Regulation:
- Guide the patient through exercises to practice self-regulation outside of biofeedback sessions. Encourage regular practice to enhance the effectiveness of EEG biofeedback.
Progress Monitoring – Step 9
- Regular Assessment:
- Record follow-up EEG sessions to periodically assess the patient’s progress. Adjust neurofeedback protocols based on observed changes and the patient’s feedback.
Conclusion:
EEG biofeedback is a dynamic and personalized approach to migraine management, capitalizing on the individual’s ability to modulate their brainwave patterns. By following these steps and incorporating essential neurofeedback protocols, practitioners can empower patients to actively participate in regulating factors associated with migraines. Regular monitoring, adjustment, and patient engagement are pivotal to optimizing the benefits of EEG biofeedback in the long term.
These biofeedback modalities collectively provide a range of tools for individuals seeking non-pharmacological approaches to managing and potentially preventing migraines. Customized interventions, when guided by a healthcare professional, can empower individuals to take an active role in migraine management. These interventions offer a comprehensive approach to managing migraines effectively by addressing both physiological and psychological factors.
How to choose from all various biofeedback modalities the right one
Choosing the most suitable biofeedback modality for managing migraines requires careful consideration of the individual patient’s needs, preferences, and the specific triggers contributing to their migraines. The effectiveness of a particular biofeedback modality can vary based on these triggers and the underlying causes of an individual’s migraines. Therefore, a personalized approach is essential for optimal results. Since migraines are a complex and heterogeneous condition, different people may experience them due to various reasons.
Consequently, a personalized approach is essential for effective management.
Thus, tailoring the choice of biofeedback modality to the specific characteristics of a person’s migraines can enhance the effectiveness of the intervention.
To illustrate, here’s a breakdown of how biofeedback modalities may be effective based on the type of migraine triggers.
Here’s a breakdown of how biofeedback modalities may be effective based on the type of migraine triggers.
Stress-Related Migraines:
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback
• Explanation: Stress can lead to dysregulation of the autonomic nervous system. In this context, HRV biofeedback plays a crucial role by focusing on restoring balance between the sympathetic and parasympathetic branches of the autonomic nervous system. Consequently, it promotes relaxation and helps reduce stress, effectively addressing the imbalance caused by stress.
Muscular Tension-Related Migraines:
• Biofeedback Modality: EMG (Electromyographic) Biofeedback
• Explanation: Muscle tension, especially in the neck and shoulders, is a common trigger for migraines. EMG biofeedback helps individuals become aware of and control muscle tension, promoting relaxation and reducing the likelihood of tension-related migraines.
Vascular-Related Migraines:
• Biofeedback Modality: Temperature Biofeedback
• Explanation: Fluctuations in skin temperature can be linked to changes in blood vessel constriction and dilation, which may contribute to vascular-related migraines. In this regard, temperature biofeedback focuses on regulating skin temperature. As a result, this regulation can potentially impact vascular function, offering a means to manage migraines associated with vascular changes.
Emotional Arousal-Related Migraines:
• Biofeedback Modality: GSR (Galvanic Skin Response) Biofeedback
• Explanation: Emotional arousal, such as heightened stress or excitement, can trigger migraines in some individuals. In this context, GSR biofeedback measures skin conductance, offering insights into emotional states. By learning to modulate these emotional responses, individuals may better manage migraines triggered by emotional factors.
Respiration-Related Migraines:
• Biofeedback Modality: Breathing Biofeedback
• Explanation: Irregular or shallow breathing patterns can contribute to migraines. To address this, breathing biofeedback emphasizes teaching individuals controlled and rhythmic breathing techniques. As a result, this practice promotes relaxation and helps prevent migraines triggered by respiratory factors.
Hormonal Fluctuation-Related Migraines:
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback, GSR (Galvanic Skin Response) Biofeedback
• Explanation: Hormonal changes, such as those occurring during the menstrual cycle, can trigger migraines. HRV biofeedback helps regulate the autonomic nervous system, while GSR biofeedback addresses emotional arousal, providing a comprehensive approach for managing hormonal fluctuation-related migraines.
Cervicogenic Migraines (Neck-Related):
• Biofeedback Modality: EMG (Electromyographic) Biofeedback
• Explanation: Muscle tension in the neck or cervical spine may influence migraines. To address this, EMG biofeedback focuses on the neck and shoulder muscles, helping individuals become aware of and control tension in these areas. As a result, this approach may potentially reduce cervicogenic migraine triggers.
Weather-Related Migraines:
• Biofeedback Modality: Temperature Biofeedback
• Explanation: Changes in weather patterns, such as temperature or barometric pressure fluctuations, can trigger migraines in some individuals. Temperature biofeedback aims to help individuals regulate their skin temperature, potentially mitigating weather-related migraine triggers.
Visual or Auditory Stimuli-Related Migraines:
• Biofeedback Modality: GSR (Galvanic Skin Response) Biofeedback
• Explanation: Bright lights, loud noises, or other sensory stimuli can trigger migraines. In this regard, GSR biofeedback measures emotional arousal, enabling individuals to learn how to modulate their responses to visual or auditory stimuli. Consequently, this can potentially reduce the frequency of migraines.
Sleep Disturbance-Related Migraines:
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback
• Explanation: Irregular sleep patterns or inadequate sleep can contribute to migraines. To address this, HRV biofeedback promotes relaxation and stress reduction. As a result, it may help individuals improve sleep quality, potentially reducing migraine triggers related to sleep disturbances.
Menstrually Related Migraines (MRM):
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback, GSR (Galvanic Skin Response) Biofeedback
• Explanation: Hormonal and emotional factors may influence migraines linked to the menstrual cycle. HRV biofeedback addresses autonomic nervous system balance, while GSR biofeedback targets emotional arousal, offering a comprehensive approach for managing menstrually-related migraines.
Vestibular Migraines:
• Biofeedback Modality: Balance and Posture Biofeedback
• Explanation: Vestibular migraines involve symptoms such as dizziness and coordination problems. In this context, balance and posture biofeedback can assist individuals in improving their awareness of body position and movement. Therefore, this approach may help manage vestibular migraine symptoms more effectively.
Medication Overuse Headaches (MOH):
• Biofeedback Modality: Comprehensive Approach (Combining HRV, EMG, and GSR)
• Explanation: MOH can result from overusing pain medications. To address this, a comprehensive biofeedback approach targets stress, muscle tension, and emotional factors. As a result, it may help individuals reduce their reliance on medications and manage headaches more effectively.
Post-Traumatic Migraines:
• Biofeedback Modality: GSR (Galvanic Skin Response) Biofeedback
• Explanation: Migraines triggered by past trauma may have emotional components. In this case, GSR biofeedback measures emotional arousal, helping individuals modulate their emotional responses. Consequently, this can potentially reduce migraine triggers related to past trauma.
Food and Beverage-Related Migraines:
• Biofeedback Modality: GSR (Galvanic Skin Response) Biofeedback
• Explanation: Certain foods and beverages can trigger migraines. To manage this, GSR biofeedback addresses emotional arousal, helping individuals regulate their emotional responses to dietary triggers. As a result, this approach may reduce the likelihood of migraines.
Ocular Migraines:
• Biofeedback Modality: Visual Biofeedback (Notable for Neurofeedback/EEG)
• Explanation: Ocular migraines involve temporary vision loss or disturbances. Visual biofeedback, particularly neurofeedback (EEG biofeedback), may help individuals regulate brainwave patterns associated with ocular migraines.
Hypnic Headaches:
• Biofeedback Modality: Temperature Biofeedback, HRV Biofeedback
• Explanation: Hypnic headaches, also known as “alarm clock headaches,” may occur during sleep and wake individuals up. Temperature biofeedback can target vascular changes, and HRV biofeedback may help regulate autonomic responses, potentially contributing to the management of hypnic headaches.
Hormone Replacement Therapy (HRT)-Related Migraines:
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback
• Explanation: Migraines associated with hormone replacement therapy may benefit from HRV biofeedback, which can help regulate autonomic responses and mitigate hormonal triggers.
Exertional Headaches:
• Biofeedback Modality: HRV (Heart Rate Variability) Biofeedback, Temperature Biofeedback
• Explanation: Exertional headaches triggered by physical activity may be influenced by autonomic responses and vascular changes. HRV biofeedback addresses autonomic balance, while temperature biofeedback can impact vascular function. Together, these modalities offer a multifaceted approach to managing exertional headaches by targeting both autonomic and vascular components.
Psychogenic Headaches:
• Biofeedback Modality: GSR (Galvanic Skin Response) Biofeedback
• Explanation: Psychogenic headaches with a strong emotional component may benefit from GSR biofeedback, allowing individuals to manage emotional arousal and potentially reduce the impact of psychological triggers.
Chronic Paroxysmal Hemicrania (CPH):
• Biofeedback Modality: EMG (Electromyographic) Biofeedback
• Explanation: CPH, a rare headache disorder, involves severe, short-lived headaches. EMG biofeedback can help address muscle tension, potentially reducing the intensity and frequency of headaches.
Post-Concussion Headaches:
• Biofeedback Modality: Comprehensive Approach (Combining HRV, EMG, GSR)
• Explanation: Post-concussion headaches can be triggered by multiple factors, including stress, muscle tension, and emotional factors. A comprehensive biofeedback approach, which addresses these various components, may help manage post-concussion headaches more effectively.
Tension-Type Headaches:
• Biofeedback Modality: EMG (Electromyographic) Biofeedback
• Explanation: Tension-type headaches, characterized by a persistent band-like pressure, often involve muscle tension. EMG biofeedback specifically targets the recognition and control of muscle tension, potentially reducing both the frequency and severity of tension-type headaches.
Chronic Tension-Type Headaches:
• Biofeedback Modality: Comprehensive Approach (Combining HRV, EMG, GSR)
• Explanation: Chronic tension-type headaches may benefit from a comprehensive biofeedback approach, addressing stress, muscle tension, and emotional factors to provide holistic headache management.
Photophobia-Triggered Migraines:
• Biofeedback Modality: Visual Biofeedback (Notable for Neurofeedback/EEG)
• Explanation: Migraines triggered by sensitivity to light (photophobia) may find relief through visual biofeedback, particularly neurofeedback (EEG biofeedback), which can assist in regulating brainwave patterns associated with sensitivity to light.
The effectiveness of biofeedback modalities can vary among individuals, and a personalized approach, possibly combining different modalities, may be most beneficial for comprehensive migraine management. Combining different biofeedback modalities, such as HRV for stress and EMG for muscle tension, may offer a more comprehensive approach to migraine management. This approach recognizes the multifaceted nature of migraines and addresses multiple contributing factors simultaneously. For example, it is adequate to combine HRV and GSR Biofeedback in stress-related migraines. Before determining the optimal combination, it is crucial to identify the key contributors to an individual’s migraines. Stress, muscle tension, autonomic dysregulation, and physiological imbalances are common factors.
LIST OF REFERENCES
- Andrasik, F., Blanchard, E. et al. (1984). Biofeedback and relaxation training for chronic headache: A controlled comparison of booster treatments and regular contacts for long-term maintenance. Journal of Consulting and Clinical Psychology, 52(4), 609-615.
- Andrasik, F. (2007). Biofeedback in headache: An overview of approaches and evidence. Cleveland Clinic Journal of Medicine, 74(Suppl 2), S33-S38.
- Busch, V., Gaul, C., et al. (2006). Biofeedback for tension-type headache: A systematic review and meta-analysis of randomized controlled trials. European Journal of Neurology, 13(5), 382-387.
- Dybvik, T., & Furnes, B. (1999). Tension-type headache: Biofeedback, relaxation, and telephone calls as adjunctive treatments. Headache: The Journal of Head and Face Pain, 39(3), 163-170.
- Grazzi, L., Andrasik, F., et al. (2006). Headache and facial pain: Differential diagnosis and treatment. In Advances and Technical Standards in Neurosurgery (Vol. 31, pp. 3-25). Springer.
- Guan, Y., Deng, H., et al. (2020). Effectiveness of Biofeedback-Assisted Relaxation for Migraine Relief: A Randomized Controlled Trial. Pain Medicine, 21(3), 493-500.
- Nestoriuc, Y., Martin, A., et al. (2008). Biofeedback treatment for headache disorders: A comprehensive efficacy review. Applied Psychophysiology and Biofeedback, 33(3), 125-140.
- Penzien, D. B., & Andrasik, F. (2004). Behavioral management of recurrent headache: Three decades of experience and empiricism. Applied Psychophysiology and Biofeedback, 29(1), 2-21.
- Ros, T., Munneke, M. A. M., et al. (2010). Endogenous control of waking brain rhythms induces neuroplasticity in humans. European Journal of Neuroscience, 31(4), 770-778.
- Sorbi, M. J., Mak, S. B., et al. (2006). Pulse rate variability is not a valid indicator for sympathetic tone during a tilt test in migraine patients. Autonomic Neuroscience, 126-127, 213-219.