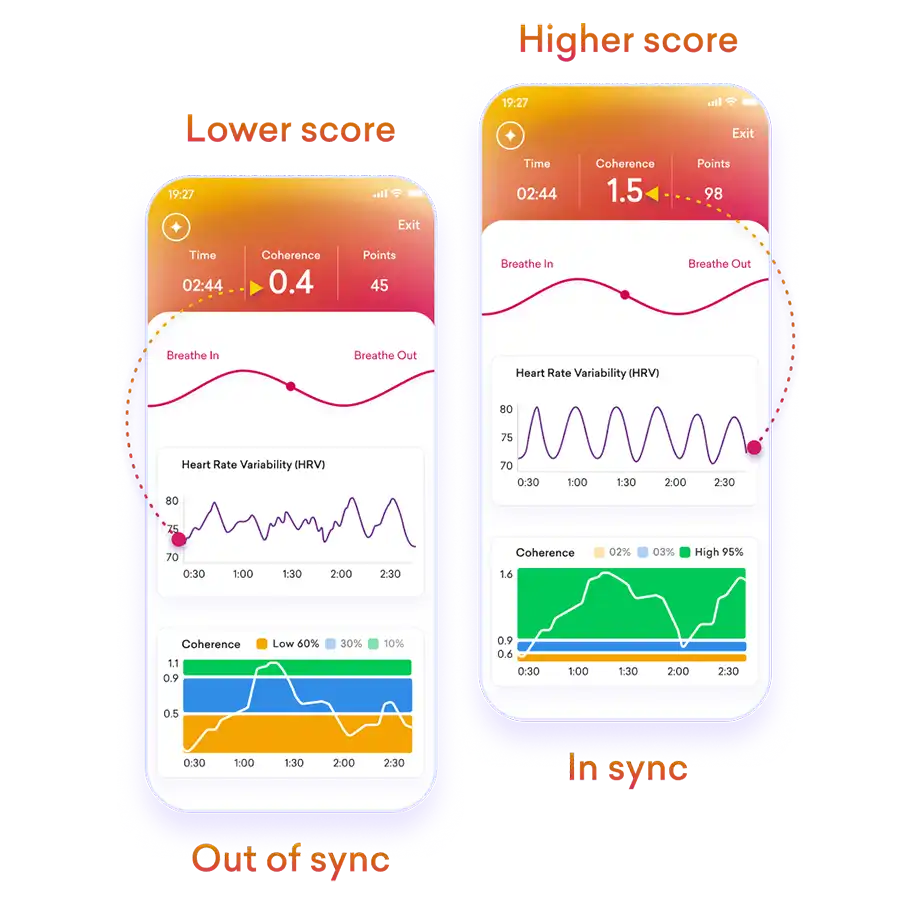
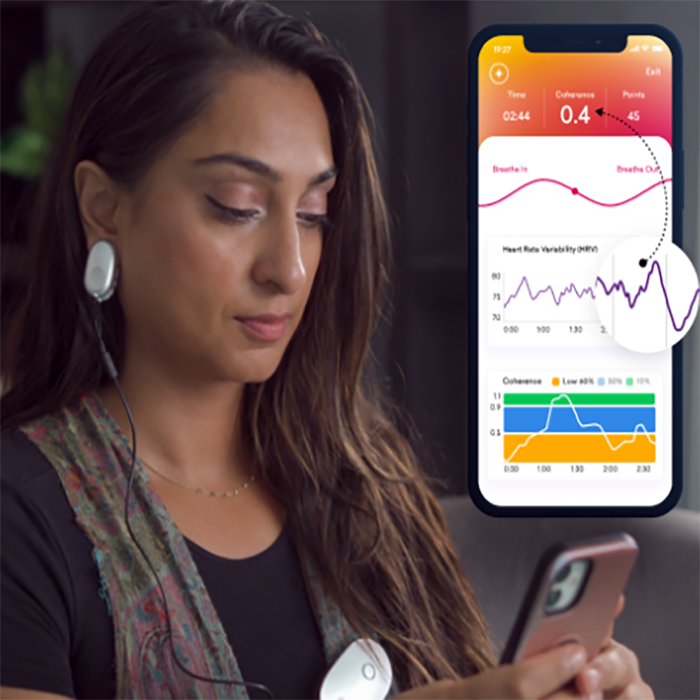
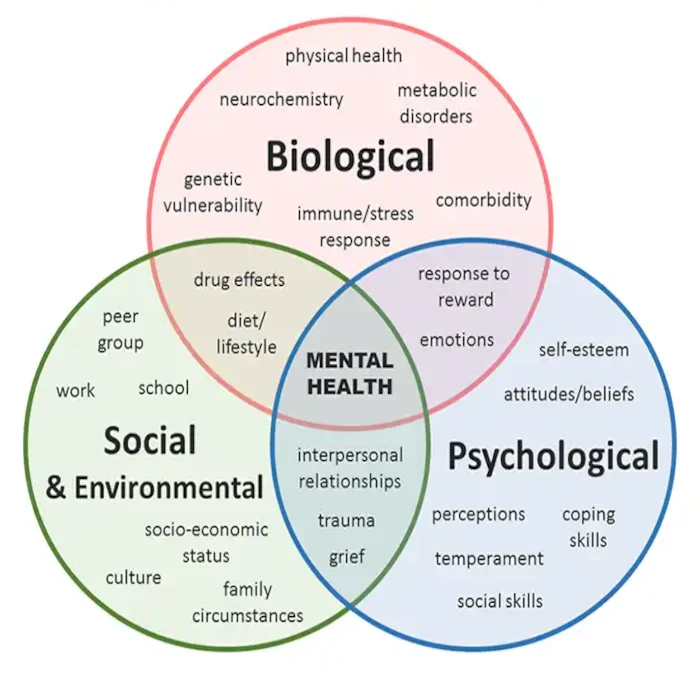
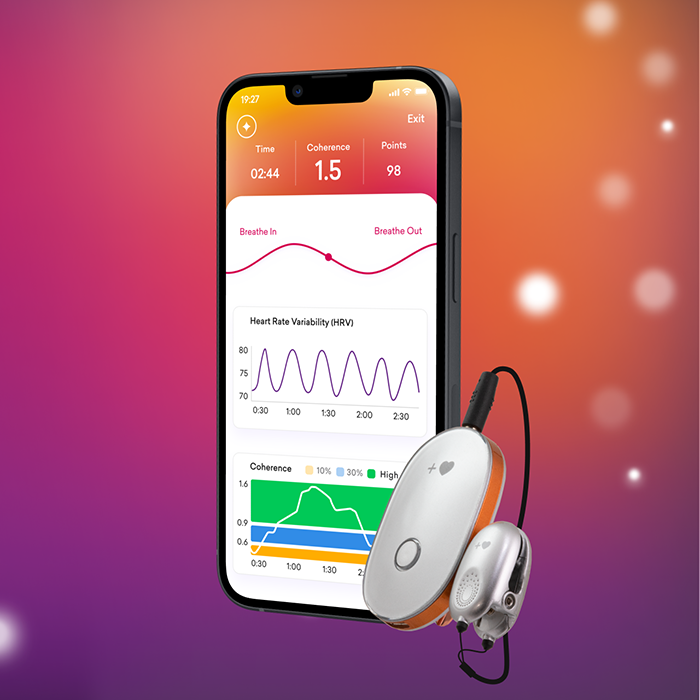
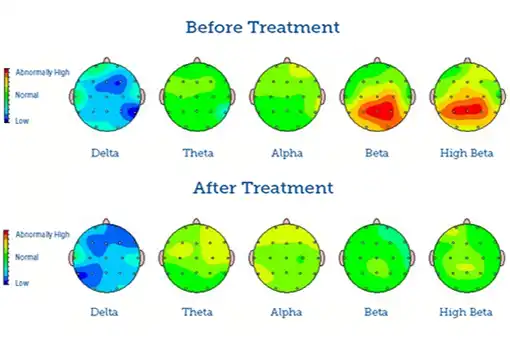
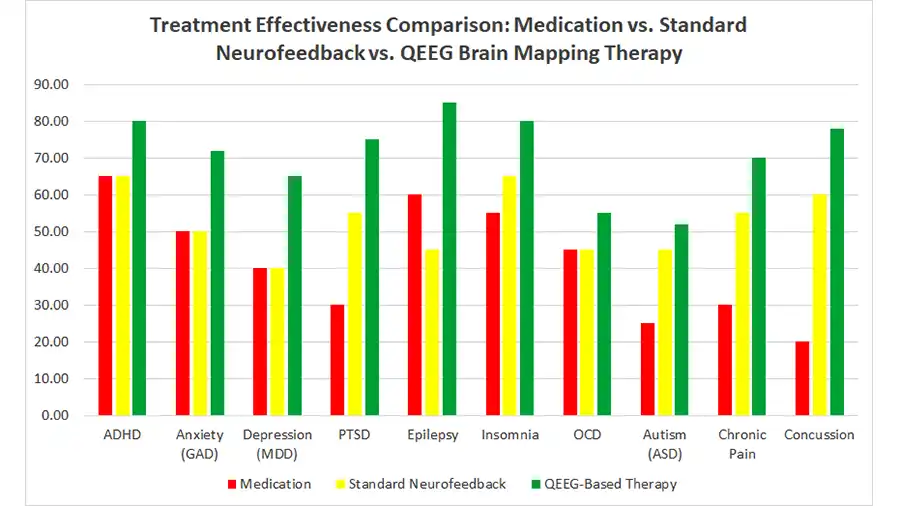
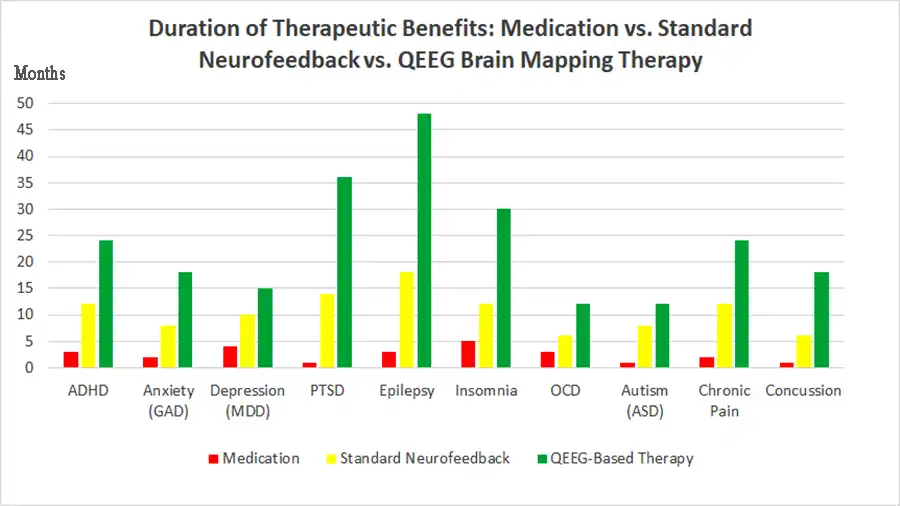
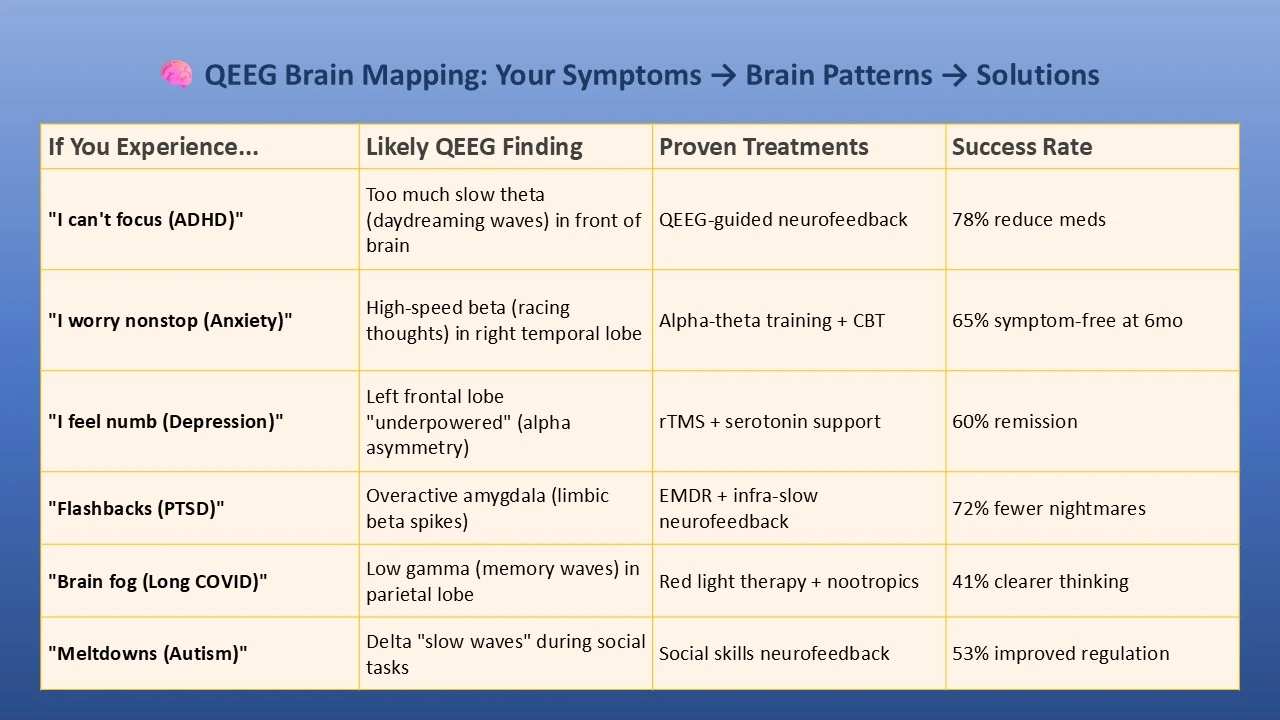
Do you feel stuck thinking the same way, struggling to learn new skills, or forgetting important details? It’s not just frustration—it’s a sign your cognitive flexibility, cognitive learning, flexibility in thinking, and working memory could use a boost. Imagine being able to switch perspectives effortlessly, absorb new information faster, solve problems creatively, and remember key details when you need them most. With the right training, these essential mental skills aren’t fixed—they can grow, adapt, and transform how you think, learn, and perform every day.
Boost Cognitive Flexibility with CogniFit
Do you ever notice you keep solving problems the same way—even when that approach fails? That’s a common sign your brain needs more cognitive flexibility. When your mind gets stuck in repetitive patterns, small obstacles feel huge, learning slows down, and everyday decisions become draining. The good news: this is fixable.
Many people confuse raw knowledge with mental agility. You can know a lot and still struggle to adapt. That’s because true mental agility relies on several interacting skills: working memory to hold information in mind, effective cognitive learning strategies to integrate new patterns, and the ability to switch mental sets—often called cognitive flexibility. Strengthening these areas helps you solve problems faster, stay calm under pressure, and learn new things more effectively.
In this article, I’ll show you a clear path from problem to solution. First, we’ll explain what cognitive flexibility really is and why it matters. Then we’ll unpack the science behind flexibility in thinking, and how cognitive learning and working memory support it. Finally, you’ll get practical, tested exercises and learn how CogniFit’s targeted training tools can accelerate progress—without guesswork.
If you’re thinking, “That’s my problem — I get stuck and can’t pivot,” you’re in the right place. Read on: you’ll find bite-sized explanations, quick exercises you can use today, and a realistic plan to build mental flexibility with CogniFit.
What Is Cognitive Flexibility?
If you’ve ever felt mentally “locked in,” repeating the same thoughts or struggling to switch tasks smoothly, you’ve experienced low cognitive flexibility. It’s not a personal flaw—it’s how the brain behaves when it isn’t trained to adapt. Cognitive flexibility is your mind’s ability to shift perspectives, adjust to new rules, change strategies, and respond creatively when situations change.
Think of it this way: life constantly throws curveballs. Plans shift, problems evolve, and technologies update. People react differently than expected. When your brain isn’t flexible, these changes feel overwhelming. Stress increases, decision-making slows, and learning new skills takes far longer than it should.
However, when cognitive flexibility is substantial, everything feels lighter. You can switch from one idea to another without friction. But you stay calm when something goes wrong. You solve problems faster because you’re not stuck using old methods. Also, you process new information more efficiently, which facilitates cognitive learning and makes it more enjoyable.
Most importantly, improving cognitive flexibility is not just for students or professionals. It supports better relationships, stronger emotional regulation, and even improved mental health. And because your brain can rewire itself throughout life, you can train this skill at any age.
The Science Behind Flexibility in Thinking
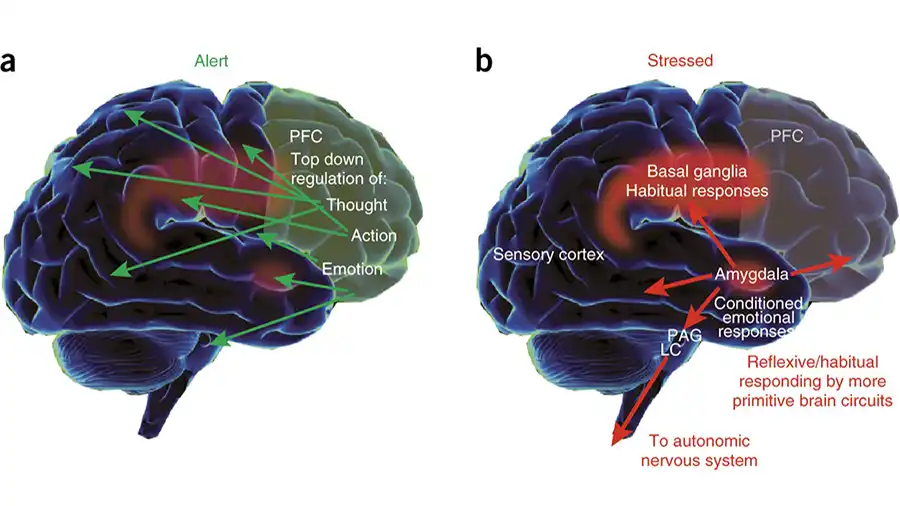
Have you ever wondered why some people adjust instantly when plans change, while others feel mentally “frozen”? The answer lies in how the brain manages cognitive flexibility, a skill mediated by communication among several key brain regions—especially the prefrontal cortex.
Your brain constantly receives new information. To stay adaptive, it must update, reorganize, and let go of old patterns quickly. When this system works well, you can switch ideas, actions, or perspectives without stress. But when it slows down, you get stuck repeating the same responses even when they no longer work.
Three mental processes shape flexibility in thinking:
1. Cognitive Flexibility
This is your ability to shift gears mentally. When cognitive flexibility is low, even small changes feel overwhelming. When it’s strong, you can pivot smoothly and solve problems with ease.
2. Working Memory
Working memory helps you hold and manipulate information while you think. It allows you to consider alternatives, compare solutions, and maintain cognitive organization. Weak working memory makes flexible thinking much harder.
3. Cognitive Learning
The brain continually forms new patterns through cognitive learning. The more efficiently you learn, the faster you can replace old mental habits with updated ones. This supports creative, adaptable thinking.
When all three work together, you become more resilient, more creative, and far better at navigating everyday challenges. And because the brain is highly trainable, you can strengthen each of these abilities through targeted cognitive exercises—something we’ll explore in the next chapters.
How Cognitive Learning Impacts Your Brain
If you’ve ever felt like learning new skills takes too long—or that information “slips away” right when you need it—then your cognitive learning system is probably working harder than it should. Many people think learning is primarily about memorizing facts, but authentic cognitive learning is far deeper. It’s the brain’s ability to absorb, connect, and apply new information in a meaningful way.
When cognitive learning functions well, your brain forms new pathways efficiently. You understand concepts faster, recall them more easily, and adapt them to new situations. But when this system slows down, everything becomes more difficult: learning new software, understanding instructions, or even remembering details from conversations.
Here’s how cognitive learning shapes your daily life:
1. It strengthens working memory
As you learn, your working memory continually updates. It stores information temporarily, enabling you to compare ideas, solve problems, and understand next steps. Poor working memory makes learning feel like “one step forward, two steps back.”
2. It boosts cognitive flexibility
The more your brain learns, the easier it is to adjust strategies when circumstances change. Strong cognitive learning lays the foundation for greater cognitive flexibility, enabling you to adapt rather than get stuck in old patterns.
3. It supports flexibility in thinking
Good cognitive learning habits help you form new mental frameworks. This directly enhances your thinking flexibility, making it easier to view problems from different angles and develop creative solutions.
The best part? Cognitive learning can improve at any age with proper training. Structured programs—such as those in CogniFit—guide your brain through exercises that strengthen these systems together, not separately. That’s how real, lasting improvement happens.
The Role of Working Memory in Daily Life
Do you ever walk into a room and forget why you went there? Or start a task, get interrupted, and lose your entire train of thought? These everyday frustrations often point to one core issue: weak working memory.
Working memory is your brain’s “mental workspace.” It stores information temporarily while you think, plan, make decisions, or solve problems. When working memory is strained, even simple tasks can feel scattered or overwhelming. You may know precisely what you want to do, but your mind can’t hold the steps long enough to complete them smoothly.
Here’s how working memory shapes your daily life:
It keeps your thoughts organized
Working memory helps you juggle ideas without losing track. When it’s strong, you can plan your day, multitask, or follow multi-step instructions without confusion. When it’s weak, your thoughts feel fragmented—you know the beginning but lose the middle or the end.
It enhances cognitive learning
Each time you learn something new, your working memory holds important details until they are transferred to long-term memory. The stronger your working memory, the more efficient your cognitive learning becomes. You don’t just remember—you understand.
It supports cognitive flexibility
You can’t shift perspectives or change strategies if your mind can’t hold alternative ideas long enough to compare them. This is why building cognitive flexibility is nearly impossible without first strengthening working memory.
It enables flexibility in thinking
When your brain can store and manipulate ideas easily, you become more adaptable. You can identify additional solutions, evaluate them clearly, and pivot when something unexpected arises. Strong working memory is the hidden engine behind true flexibility in thinking.
The encouraging part is that working memory is highly trainable. With regular, targeted cognitive exercises—such as those offered by CogniFit—you can significantly strengthen this mental system. Better working memory leads to clearer thinking, faster learning, and a more agile mind overall.
How CogniFit Enhances Cognitive Flexibility
If you’ve ever tried random brain games hoping they would improve your thinking, you’re not alone. Many people expect that simple puzzles or apps will sharpen their minds—but most tools don’t target the specific skills that actually drive cognitive flexibility, cognitive learning, flexibility in thinking, and working memory. CogniFit is different because it uses personalized, science-backed training that adapts to your brain’s needs.
CogniFit starts by assessing your cognitive profile. This isn’t a generic quiz. It’s a clinically validated evaluation used in research, healthcare, and education. The system identifies where you struggle most—perhaps switching tasks, remembering details, or adapting to new information. It then develops a customized training program to address those specific weaknesses.
Here’s how CogniFit improves the core skills behind flexible and adaptive thinking:
Boosting Working Memory
Many CogniFit exercises challenge you to hold and update information in real time. This strengthens the mental workspace that supports every higher-level cognitive skill. As working memory improves, you think more clearly and react more confidently.
Enhancing Cognitive Learning
CogniFit engages multiple learning systems simultaneously—pattern recognition, emotion processing, problem solving, and attention. This speeds up cognitive learning, helping you absorb new ideas more efficiently and apply them with less effort.
Improving Flexibility in Thinking
By combining memory, attention, and reasoning tasks, CogniFit stimulates neural pathways that support cognitive flexibility. With consistent training, your brain becomes better at generating solutions, adapting to change, and thinking creatively.
What makes CogniFit especially effective is its adaptive difficulty. As you get better, the challenges evolve. You’re constantly training at the sweet spot—complex enough to grow but not so difficult that you feel discouraged. This ensures steady improvement, supported by measurable results that can be tracked over time.
Exercises to Improve Cognitive Learning
If you want to learn faster, remember more, and understand complex ideas with less effort, you must train your cognitive learning system. The good news? You don’t need hours of studying or complicated tools. Simple, consistent exercises—paired with CogniFit’s personalized brain training—can dramatically improve how efficiently your mind absorbs and applies new information.
Here are practical exercises you can start using right away:
1. Pattern Expansion Drills
Your brain loves patterns. When you expand, rearrange, or predict sequences, you strengthen neural pathways involved in cognitive learning.
Try this:
Start with a simple number or letter pattern and increase its complexity incrementally. Your brain becomes quicker at detecting structure and meaning.
2. Story Reconstruction
Tell yourself a short story, wait one minute, then retell it in your own words. This builds working memory and strengthens how your brain processes new information. You don’t just memorize—you learn to understand and reinterpret.
3. Flexibility Challenges
Practice explaining the same idea in three different ways. This enhances flexibility in thinking and trains the cognitive system to approach information from multiple perspectives. The more viewpoints you can generate, the stronger your learning becomes.
4. Switching Tasks Mindfully
Set a timer and switch between two unrelated tasks every 60–90 seconds. This trains cognitive flexibility, helping your mind stay adaptable while learning new material.
5. CogniFit Cognitive Training
CogniFit integrates these skills into a single adaptive system. Its exercises automatically adjust to your skill level, pushing your cognitive learning capacity without overwhelming you. This structured approach helps you progress faster than with random brain games or unstructured practice.
These exercises build the cognitive foundations necessary for rapid, efficient learning. When paired with CogniFit’s science-based training, your progress becomes measurable and easier to maintain over time.
Training Flexibility in Thinking with CogniFit
If you’ve ever caught yourself saying, “I just can’t think of another way to solve this,” then you’ve experienced a drop in flexibility in thinking. This mental rigidity shows up in everyday life—struggling to adapt when plans change, getting frustrated when problems don’t have obvious answers, or feeling mentally “boxed in” during conversations or challenges. The reassuring news? You can train your brain to break free from these rigid patterns.
Flexibility in thinking depends on your brain’s ability to shift perspectives, consider alternatives, and update old mental habits. When this skill is weak, your mind gravitates to the exact solutions over and over, even if they don’t work. When it’s strong, you become more creative, adaptable, and emotionally resilient.
Here are powerful techniques to enhance your flexibility in thinking, especially when paired with CogniFit’s structured cognitive training:

Perspective Reversal
Pick a belief or assumption and flip it completely.
Ask yourself: What if the opposite were true?
This disrupts mental rigidity and teaches your brain to explore new angles.
Alternative Route Mapping
Take a simple problem—like planning your day or completing a task—and write down three completely different ways to do it. This boosts your cognitive flexibility and forces your mind to consider multiple pathways.
Idea Expansion Exercises
Choose an object or concept and list as many uses or interpretations as possible. The more you stretch your creative thinking, the more flexible your cognitive processes become.
Working Memory Stacking
Try holding several ideas in your mind at once and compare them. This strengthens working memory, which is essential for keeping multiple options “active” while you think through them.
CogniFit’s Adaptive Thinking Tasks
CogniFit integrates all these skills into its training system. Its exercises require you to shift rules, update strategies, and adjust quickly. Because the difficulty adapts to your performance, your brain stays challenged at the perfect level—helping you build long-lasting flexibility in thinking. Over time, this makes you faster at problem-solving, better at creative tasks, and more emotionally adaptable.
Training flexibility in thinking doesn’t just improve your intellectual performance—it transforms how you react to life. You respond rather than freeze, and you adapt rather than resist. Also, you think clearly instead of feeling overwhelmed.
Boost Working Memory Through Targeted Brain Games
If you’ve ever felt mentally overloaded—like your thoughts scatter the moment you try to juggle more than one idea—you’re experiencing the strain of limited working memory. This skill is the backbone of clear thinking. Without strong working memory, it becomes harder to plan, reason, learn, or stay focused. The good news is that working memory is highly trainable, especially with structured tools like CogniFit.
Working memory determines how well your brain can hold information, manipulate it, and use it in real time. When it functions well, you think faster, stay organized, and solve problems with ease. When it’s weak, your mind feels cluttered or chaotic—even if you’re intelligent and motivated.
Here’s how you can strengthen your working memory through targeted exercises and CogniFit’s adaptive training:
Dual-Task Challenges
Try performing two simple tasks at once—like reciting numbers while sorting objects by size or color. This forces your working memory to manage multiple streams of information. CogniFit includes dual-task exercises designed to safely and effectively challenge this skill.
Sequence Expansion
Pick a sequence (numbers, shapes, movements) and gradually increase its length. Each step challenges your working memory to stretch a little further. This is the foundation of many CogniFit brain games that scale to your ability.
Mental Rotation Tasks
Visualize an object and rotate it in your mind. This kind of mental manipulation strengthens the “processing” side of working memory, not just storage. CogniFit uses these tasks to improve spatial reasoning and mental agility.
Cognitive Flexibility Switch Tasks
Switch between rules—like “sort by color” and then “sort by size.” This strengthens both cognitive flexibility and working memory, since your brain must hold the rules while shifting between them.
CogniFit’s Adaptive Memory Training
CogniFit personalizes its working memory exercises based on your performance. If you struggle in a particular area, the system delivers more targeted tasks. If you excel, it gently raises the difficulty. This ensures continuous growth, which is impossible with fixed-level games or simple memory apps.
When your working memory grows stronger, something amazing happens:
- Your mind feels clearer.
- Learning becomes easier.
- And your flexibility in thinking improves naturally—because your brain can now manage more information at once.
Measuring Your Progress: Tracking Cognitive Gains
One of the biggest challenges in self-improvement is knowing whether your efforts are actually working. When it comes to building mental agility, the changes can feel subtle day to day. That’s why tracking your cognitive progress—especially improvements in cognitive flexibility, cognitive learning, flexibility in thinking, and working memory—is essential. Precise measurements help you stay motivated, see real results, and understand which areas still need attention.
CogniFit makes this process simple. Its platform doesn’t just give you exercises—it provides data-driven insights that show how your brain is evolving. Instead of guessing or hoping you’re improving, you see measurable cognitive gains over time.
Here’s how to track your progress effectively:
Baseline Evaluation
Before training, CogniFit runs a comprehensive cognitive assessment. This maps your strengths and weaknesses across dozens of skills, including working memory, cognitive flexibility, reasoning, attention, and processing speed. Your baseline allows you to compare future results with your starting point.
Skill-Specific Scores
As you train, CogniFit updates your scores for targeted abilities. You’ll see which cognitive skills improve fastest and which ones need more focused training. This is especially valuable for monitoring progress in cognitive learning and flexibility in thinking.
Progress Graphs & Trends
CogniFit’s dashboard displays visual trends showing how your performance shifts over days and weeks. These graphs help you understand whether your training intensity is right and how consistently you’re improving.
Real-Life Feedback
Numbers matter—but your daily life gives you the most evident signs of growth.
You’ll notice you:
- switch tasks with less frustration (stronger cognitive flexibility)
- remember details more easily (improved working memory)
- learn new skills faster (better cognitive learning)
- find creative solutions where you once felt stuck (enhanced flexibility in thinking)
Adjusting Your Training Plan
CogniFit adapts automatically, but you can also adjust your schedule or focus areas. If you feel mentally rigid or overwhelmed, you might emphasize cognitive flexibility tasks. If learning feels slow, boosting working memory can help.
Tracking your progress isn’t just data—it’s motivation. When you see your mind become clearer, faster, and more adaptable, you reinforce the habit of consistent training. And that momentum leads to even bigger improvements.
FAQ: Boost Cognitive Flexibility in Thinking
Cognitive flexibility is the brain’s ability to switch between thinking patterns, adapt to new situations, and consider multiple perspectives. Improving cognitive flexibility helps you solve problems faster, manage stress, and enhance flexibility in thinking, making learning and decision-making easier.
Cognitive learning involves absorbing, understanding, and applying new information efficiently. You can enhance it through targeted exercises like pattern recognition, memory challenges, and brain games. Programs like CogniFit offer structured exercises that train cognitive skills while boosting working memory and cognitive flexibility.
Flexibility in thinking refers to your ability to view problems from different angles and generate multiple solutions. While closely related to cognitive flexibility, which emphasizes mental switching, flexibility in thinking focuses more on creative and adaptive problem-solving. Training both skills together strengthens overall brain agility.
Working memory is your brain’s temporary storage for information you actively use while thinking, learning, or making decisions. Strong working memory supports cognitive learning and enhances both cognitive flexibility and flexibility in thinking, enabling you to manage tasks more effectively.
Results vary depending on how consistently you train, but many users notice improvements in focus, memory, and adaptability within a few weeks. Since CogniFit tracks progress over time, you can see measurable gains in cognitive flexibility, working memory, and other cognitive skills.
References
- Cognitive Flexibility Training Improves Extinction Retention Memory and Enhances Cortical Dopamine — https://pubmed.ncbi.nlm.nih.gov/30881293/ PubMed
- A Hierarchical Model of Cognitive Flexibility in Children — https://pubmed.ncbi.nlm.nih.gov/31913075/ PubMed
- Comparing Working Memory Capacity with Cognitive Flexibility and Learning Styles — https://karger.com/article/doi/10.1159/000526226 Karger Publishers
- Cogmed Cognitive Training for Working Memory: Meta-analysis — https://pubmed.ncbi.nlm.nih.gov/40581145/ PubMed
- Combined Exercise and Cognitive Training Improves Working Memory in Older Adults — https://pubmed.ncbi.nlm.nih.gov/37065695/ PubMed